Technological Applications of Platinum
Catalyst

A catalyst is something that helps speed up a reaction but is not consumed itself. Platinum has many uses in industries. It helps form petroleum and petroleum products from crude oil. Petroleum products are things such as gasoline, plastics, and skin products. Platinum is also used to form fertilizers, synthetic fibres (such as rayon), drugs, vitamins, antibiotics, hydrogen peroxide, hydrogen cyanide, and sulfuric acid. Platinum helps form sulfuric acid by causing oxygen to combine with sulfur dioxide to form sulfur trioxide, which is an important step in the process. Ground up platinum, or platinum black, is more efficient than its larger form because of the increased surface area.
Catalytic Converter

Platinum is a very stable metal, which makes it a useful material for many objects. one of its most important uses is for a catalytic converter, which was created in 1975. A catalytic converter is located near the exhaust on a car. It helps the gasoline combust more completely. It reduces the amount of harmful emmisions in the air by using air to convert them to less harmful substanes. Carbon monoxide, hydrocarbons, and nitrous oxide are converted into carbon dioxide, water, and nitrogen gas.
Jewelry

Platinum is ideal for jewelry because it is as unreactive as gold. It is also more expensive than gold and can be used to make an alloy, called platinum gold. This alloy looks like silver or white gold. Platinum is very ductile and malleable, so it can be shaped into many forms. it is a beautiful setting for diamonds and makes a popular engagement ring. Platinum is used to make rings, necklaces, bracelets, earrings, piercings, pins, etc.
Crucibles

A crucible is a container that is used to bring substances to very high temperatures. Platinum has a very high melting point and is unreactive with most substances, which make it ideal for laboratories. A platinum crucible must not be used when melting alkaline substances, as hot, strong bases can dissolve it.
Cancer Treatments
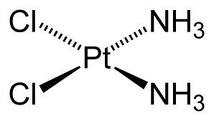
Two platinum-based anti-cancer drugs are currently on the market, cisplatin (pictured) and carboplatin. Both may have toxic side effects. It was difficult to produce aqueous solubility since platinum is not soluble in water.
Pacemakers
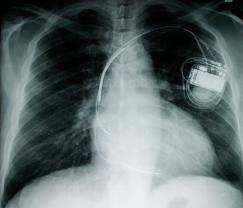
Platinum is an ideal material to create pacemaker electrodes from because it conducts electricity and does not dissolve form the body's acids. A pacemaker is used when someone's heart is unable to create its own steady pace. The platinum electrodes will deliver electrical impulses to the heart muscles.
Magnets

Platinum itself is not very magnetic, but when alloyed with cobalt, one of the strongest magnets is formed. These magnets can be used from electromagnets to wristwatches.
Platinum Resistance Thermometre

Like all thermometres, a platinum resistance thermometre is used to determine temperature. The pure platinum of which it is composed measures the resistance travelling through it. It works because the resistance of metals are proportional to the temperature (meaning they produce a linear graph). The resistance and volt are used in an equation to convert to temperature.
